|
This background can come from non-specific binding during antibody incubation, membrane background in the case of fluorescent images, or background arising from the instrumentation itself.įor bright, isolated bands on smooth backgrounds, this process can be relatively straightforward. No western blot is perfect, and all blot images show some level of background. 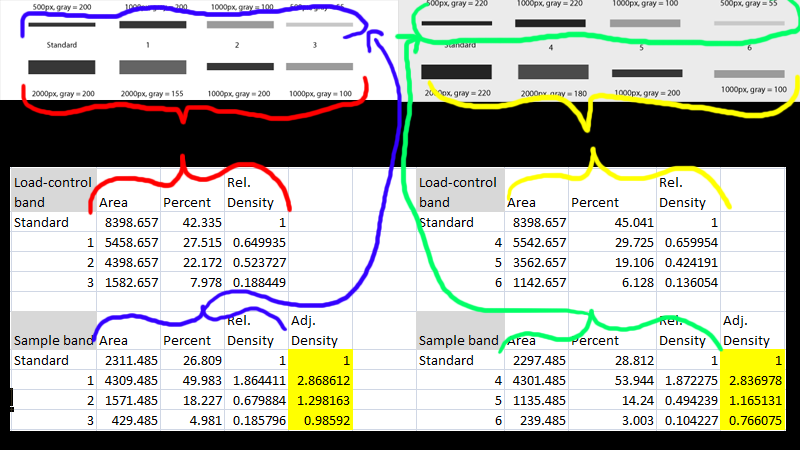
Therefore, band quantity is often referred to as band "volume" by imaging software. Each pixel also carries signal intensity information, which can be thought of as data in the third dimension. The pixels that comprise the part of a band have x and y coordinates, so outline an area. Therefore, band quantity can be thought of as a volume, which is the total amount of signal for all the pixels within that band. Each pixel in a blot image has an x and y coordinate, in addition to an intensity value, which occupies the third dimension. A digital image of a blot can be thought of as data in three dimensions. A western blot image is made up of pixels, which contain information about how much signal was collected at each location in the image. Band VolumeĪ protein band is a feature that appears in a western blot image. Note: western blots are semi-quantitative so they are ideal for showing relative protein expression rather than an absolute quantity. By analyzing the intensity of the signal, you may determine whether the expression of the target protein in one sample has increased or decreased relative to another sample or control. The signal intensity of the band is directly proportional to the concentration of your target protein. Quantitating a western blot refers to the measurement of the signal emitted by your protein band(s) of interest. In the end, the integrity and robustness of the published scientific data should be part of the considerations while planning image analysis experiments.Blot Quantitation and Background What Does Quantitation Mean? Sometimes, a qualitative interpretation might be better suitable than a flawed quantification. IMHO: Not because we can measure everything means also we should do it. I can only recommend to use either the software of the Western Blot imaging system in the lab or alternatively GelAnalyzer because it allows to stick pretty well to the recommended procedure by Western Blot material suppliers (which I would guess are the most experienced people in that particular field) and as seen in the publication above. Hammond, “A defined methodology for reliable quantification of Western blot data.,” Mol. So, generally there are many pitfalls related to WB measurements.īesides the literature list in one of the linked posts above, mainly the following gives a good insight into the procedure: And band selections which overshoot the actual band also lead to wrong results. One cannot reliably quantify bands by making boxes around them and using analyze measure nor do horizontal lines fulfill the requirements. Here is one video which is explaining a little more detailed the considerations of WB in general, normalization as well as measurements 
The latter and pretty much of most other ones completely ignore every thing mentioned in scientific literature regarding Western Blot measurements (and the pre-requisites for it). There are many videos online like the linked one above. But I cannot contain myself to add my 2 cents to this topic, because some of those videos make me like… No offense to no one making those videos or taking them as orientation in case of the lack of other available resources. This was the one I looked at Analysing blots and gels with ImageJ/Fiji - YouTube
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
